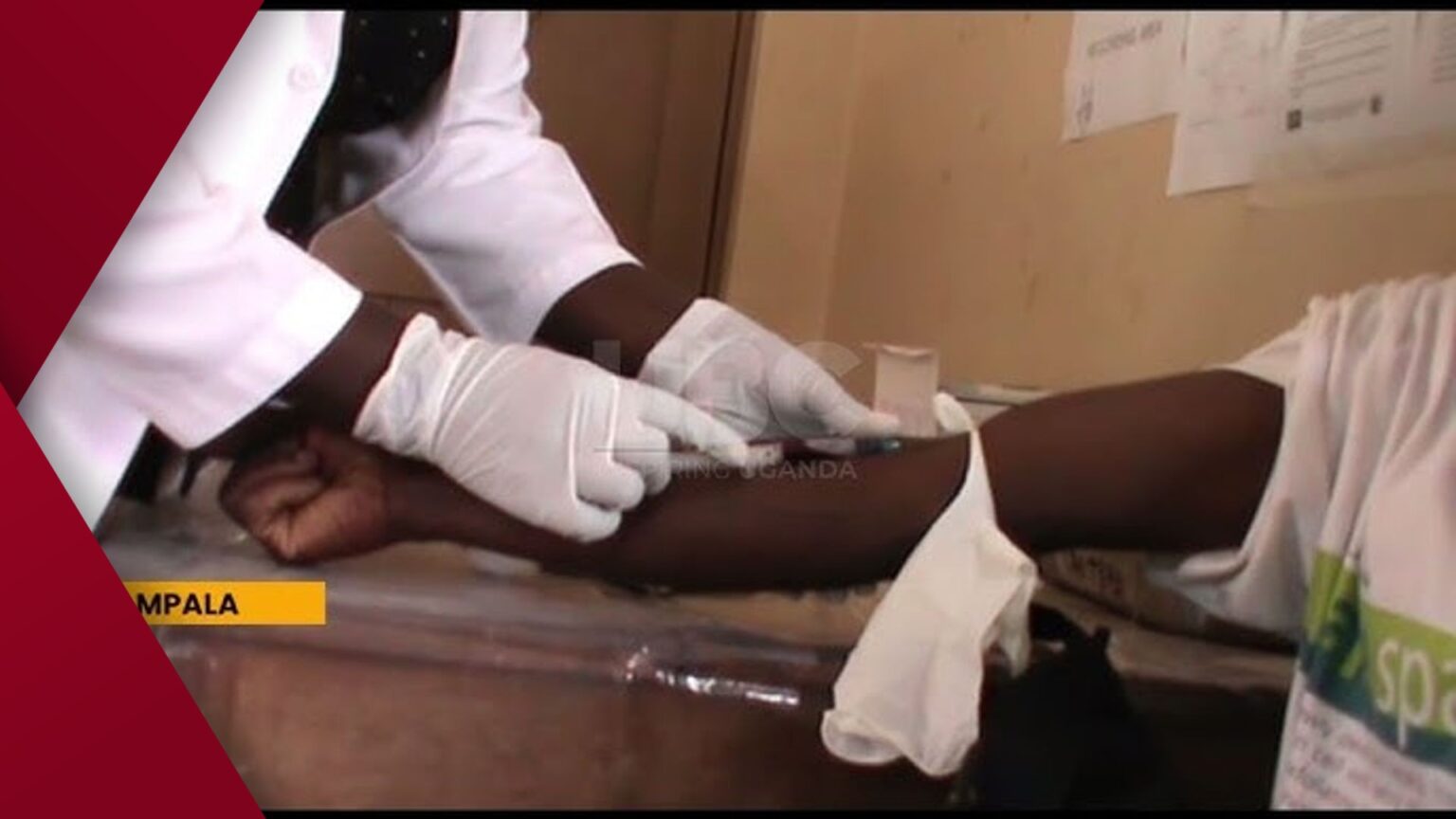
Uganda Drug Authority Approves New HIV Medicine. KAMPALA, UGANDA — The Uganda National Drug Authority (NDA) has approved Lenacapavir, a new injectable pre-exposure prophylaxis (PrEP) medication for HIV prevention, manufactured by Gilead Sciences, a pharmaceutical company based in the United States.
The approval represents a significant advancement in HIV prevention strategies within Uganda.
“This is a game-changer for HIV prevention, especially for those at high risk,” the country’s drug authority stated on 5 January.
The NDA described the approval as a substantial breakthrough that could revolutionize HIV prevention in Uganda, aiming to contribute to the goal of ending AIDS by 2030. Lenacapavir, marketed under the brand name Sunlenca, is a long-acting antiretroviral medication designed for treating and preventing HIV/AIDS.
It differs from the standard daily oral PrEP pills, offering a more convenient twice — yearly injection.
Recent clinical trials have shown that Lenacapavir can prevent more than 99 percent of new HIV infections among high-risk individuals. Uganda is among 10 high-burden countries selected to receive support under the United States President’s Emergency Plan for AIDS Relief (PEPFAR).
Efforts are underway to ensure the affordability of the new drug, with co — financing from the US government and the Global Fund for an advanced market financing arrangement.
Uganda plans to allocate approximately US$1. 14 million from its Global Fund grant to support the launch of the Lenacapavir programme.
In July 2025, the World Health Organisation (WHO) released new policy guidelines recommending Lenacapavir as a key tool for HIV prevention. WHO Director-General Dr Tedros Adhanom Ghebreyesus emphasized the drug’s potential, stating, “While an HIV vaccine remains elusive, Lenacapavir is the next best thing: a long-acting antiretroviral shown in trials to prevent almost all HIV infections among those at risk. ” The WHO’s guidelines follow a critical moment in global HIV prevention efforts, as 2024 saw an estimated 1.
3 million new HIV infections, disproportionately affecting key and priority populations. Currently, approximately 1. 5 million people are living with HIV in Uganda, underscoring the ongoing public health challenge despite progress in treatment and prevention.
Welcoming the approval, several individuals on X called for greater recognition of the role played by Ugandan scientists in the development of the PrEP.
The WHO is urging governments, donors, and global health partners to immediately integrate Lenacapavir into national combination HIV prevention programmes, emphasizing the importance of collecting data on the drug’s uptake, adherence, and real-world impact.
A number of African countries have already approved Lenacapavir and initiated implementation.
Kenya, Nigeria, Lesotho, Mozambique, Malawi, Rwanda, Tanzania, Botswana, and Namibia are either in the early adoption stages or planning national roll — outs.